
 Background: The Surviving Sepsis Campaign published their recommendations for the management of patients with COVID-19 on March 20th, 2020 (though as of the date of this blog post, the document has not been peer reviewed). 36 experts from 12 countries attempted to answer 53 questions that are relevant to the management of COVID-19 patients in the ICU. 54 recommendations were made of which 4 are best practice statements, 9 are strong recommendations, and 35 are weak recommendations. Finally, no recommendations were provided for 6 of the questions. The document is divided into 5 sections:
Background: The Surviving Sepsis Campaign published their recommendations for the management of patients with COVID-19 on March 20th, 2020 (though as of the date of this blog post, the document has not been peer reviewed). 36 experts from 12 countries attempted to answer 53 questions that are relevant to the management of COVID-19 patients in the ICU. 54 recommendations were made of which 4 are best practice statements, 9 are strong recommendations, and 35 are weak recommendations. Finally, no recommendations were provided for 6 of the questions. The document is divided into 5 sections:
- Infection Control
- Laboratory Diagnosis and Specimens
- Hemodynamic Support
- Ventilatory Support
- COVID-19 Therapy
Below is the list of recommendations and I will interject my thoughts on the ones that need them.
Paper: Alhazzani W et al. Surviving Sepsis Campaign: Guidelines on the Management of Critically Ill Adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med 2020. [Epub Ahead of Print]
Legend on Levels of Recommendation:
- Best Practice Statement = BPS
- Weak Recommendation, Very Low Quality Evidence = WR-VLQE
- Weak Recommendation, Low Quality Evidence = WR-LQE
- Weak Recommendation, Mod Quality Evidence = WR-MQE
- Strong Recommendation, Low Quality Evidence = SR-LQE
- Strong Recommendation, Mod Quality Evidence = SR-MQE
- Strong Recommendation, High Quality Evidence = SR-HQE
Infection Control:
-
Aerosol-generating procedures* = Fitted respiratory masks (N95, FFP2, or equivalent), in addition to other personal protective equipment (i.e. Gloves, gown, and eye protection, such as a face shield or safety goggles (BPS)
- *Aerosol-generating procedures include endotracheal intubation, bronchoscopy, open suctioning, administration of nebulized treatment, manual ventilation before intubation, physical proning of the patient, disconnecting the patient from the ventilator, non-invasive positive pressure ventilation, tracheostomy, and cardiopulmonary resuscitation
- Aerosol-generating procedures = In a negative pressure room (BPS)
- Although this recommendation is obvious, as the number of patients start to increase, we will simply not have enough negative pressure rooms to make this feasible. Therefore, if no negative pressure room is available, the next best thing would be a normal room, with the door closed and full PPE (fitted respiratory mask, gloves, gown, eye protection, and face shield)
- Usual care for non-ventilated COVID-19 patients = Surgical/medical masks, as opposed to respirator masks, in addition to other personal protective equipment (i.e. gloves, gown, and eye protection, such as a face shield or safety goggles) (WR-LQE)
- This is a key point. If we use fitted respirator masks on everyone, we will simply run out, if we haven’t already. We need to save these for the riskier procedures
- Ensure you have a triage process that puts patients with respiratory symptoms into a surgical mask. Then by standing 6 feet away from patients not in distress, a simple surgical mask instead of N95 is feasible. There are lots of physical exam findings you can comment on without touching a patient.
- Non-aerosol-generating procedures on mechanically ventilated (closed circuit) patients with suspected/confirmed COVID-19 = Surgical/medical masks, as opposed to respirator masks, in addition to other personal protective equipment (i.e. gloves, gown, and eye protection, such as a face shield or safety goggles) (WR-LQE)
- The authors mention one RCT reporting on seasonal influenza by means of PCR. 22.2% (50/225) nurses in medical mask group had RT-PCR confirmed coronavirus infection compared with 19.9% (44/221) in the N95 respiratory group [2]. Essentially no difference
- Endotracheal intubation = Video-guided laryngoscopy (VL), over direct laryngoscopy (DL), if available (WR-LQE)
- Intubation is one of the most dangerous procedures we will perform as it puts us at risk of being exposed to airway and respiratory secretions. Therefore, any technique that can reduce the number of attempts at endotracheal intubation, the duration of the procedure, and minimize the proximity between the operator and the patient, should be done.
- In addition, video laryngoscopy with a screen separate (i.e. away) from the laryngoscope would be preferred
- Another trick may be to take a clear trash bag and create a hood (Of course you would be in full PPE for this). You now have a barrier over the patients face from which you are working under to minimize potential aerosolization
- Endotracheal intubation = Performed by the healthcare worker who is most experienced with airway management in order to minimize the number of attempts and risk of transmission (BPS)
- This makes sense as we want to maximize the chances of first pass success
Laboratory Diagnosis and Specimens
- For intubated and mechanically ventilated adults with suspicion of COVID-19:
- Diagnostic testing = Recommend lower respiratory tract samples > upper respiratory tract (nasopharyngeal or oropharyngeal samples (WR-LQE)
- Lower respiratory samples = Recommend endotracheal aspirates > bronchial wash or bronchoalveolar lavage samples (WR-LQE)
- Bronchoalveolar lavage has a high risk of aerosolization and exposure of healthcare professionals. They go on to state that sputum induction should also be avoided to increased risk of aerosolization
- Tracheal aspirate specimens appear to carry a lower risk of aerosolization and can be obtained without disconnecting the patient from the ventilator
- Although we don’t know the true sensitivity and specificity of the SARS-CoV-2 RT-PCR test, based on limited information a single negative swab from the upper airway does not rule out COVID-19 infection. Repeated sampling from multiple sites will increase diagnostic yield
- Finally, a positive test for another respiratory virus does not rule out COVID-19, and should not delay testing if there is a high suspicion of COVID-19. This is a changing paradigm and important to realize, that a positive viral result does not rule out COVID-19 infection. A pre-release study out of Stanford showed a 23% rate of co-infection [Link is HERE]. Of the 49 positive SARS-CoV-2 results, 11 (22.4%) also had a co-infection.
Supportive Care:
- For patients in shock = Dynamic parameters (i.e. skin temperature, capillary refill time, and/or serum lactate measurement) over static parameters to assess fluid responsiveness (WR-LQE)
- Acute resuscitation of adults with suspected/confirmed COVID-19 and shock = Conservative over a liberal fluid strategy (WR-VLQE))
- This recommendation comes from patients with sepsis and ARDS data. Unfortunately, what “conservative” means was not listed
- Acute resuscitation of adults with suspected/confirmed COVID-19 and shock:
- Crystalloids > colloids (SR-MQE)
- Buffered/balanced crystalloids > unbalanced crystalloids (WR-MQE)
- Don’t use hydroxyethyl starches (SR-MQE)
- Don’t use dextrans (WR-LQE)
- Don’t use albumin for initial resuscitation (WR-MQE)
Vasoactive Agents:
- For adults with COVID-19 and shock:
- 1st line = norepinephrine (WR-LQE)
- If norepinephrine not available = Vasopressin or epinephrine as first-line (WR-LQE)
- Don’t use dopamine if norepinephrine is available (SR-HQE)
- 2nd line = Vasopressin > titrating norepinephrine dose, if target MAP cannot be achieved by norepinephrine alone (WR-MQE)
- For adults with COVID-19 and shock, we suggest titrating vasoactive agents to target a MAP of 60 – 65mmHg rather than higher MAP targets (WR-LQE)
- Cardiac dysfunction and persistent hypoperfusion despite fluid resuscitation and norepinephrine = Add dobutamine, over increasing norepinephrine dose (WR-VLQE)
- Refractory shock = Low-dose corticosteroid therapy (“shock-reversal”) > no corticosteroid therapy (WR-LQE)
- A typical corticosteroid regimen in septic shock is IV hydrocortisone 200mg/day administered either as an infusion or intermittent doses
- This is not all patients; this is refractory shock patients. There is some evidence that corticosteroids can prolong viral replication
Ventilatory Support:
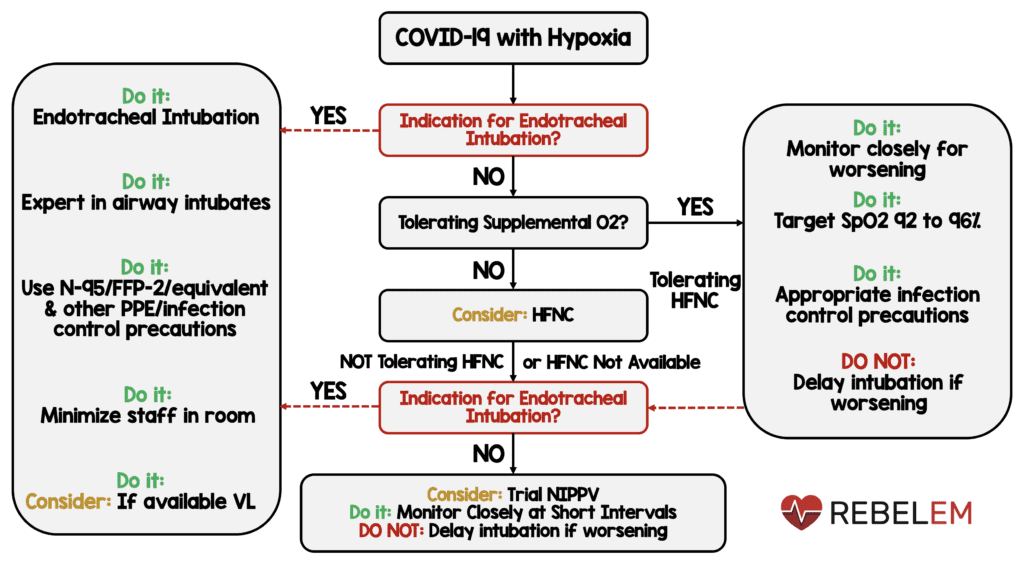
- Start supplemental oxygen if:
- SpO2 <92% (WR-LQE)
- SpO2 is <90% (SR-MQE)
- Acute hypoxemic respiratory failure on oxygen = SpO2 maintained no higher than 96% (SR-MQE)
- Acute hypoxemic respiratory failure despite conventional oxygen therapy, we suggest using HFNC over conventional oxygen therapy (WR-LQE)
- Evidence on mortality and length of stay is not as strong for HFNC, but the reduction in the need for intubation is important, when dealing with pandemics where resources such as critical care beds and ventilation may become limited
- In studies evaluating bacterial environmental contamination HFNC presented a contamination risk similar to that of conventional oxygen [3]
- An added layer of protection for me is putting these patients in a surgical mask and turning the flow down to half (i.e. 20 – 30LPM)
- Acute hypoxemic respiratory failure = HFNC > NIPPV (WR-LQE)
- Acute hypoxemic respiratory failure = If HFNC not available and no urgent indication for endotracheal intubation, a trial of NIPPV and close monitoring and short-interval assessment for worsening of respiratory failure is reasonable (WR-VLQE)
- No recommendation on helmet NIPPV vs mask NIPPV. Unclear safety and efficacy
- NIPPV or HFNC = Close monitoring for worsening of respiratory status, and early intubation in a controlled setting if worsening occurs (BPS)
Invasive Mechanical Ventilation:
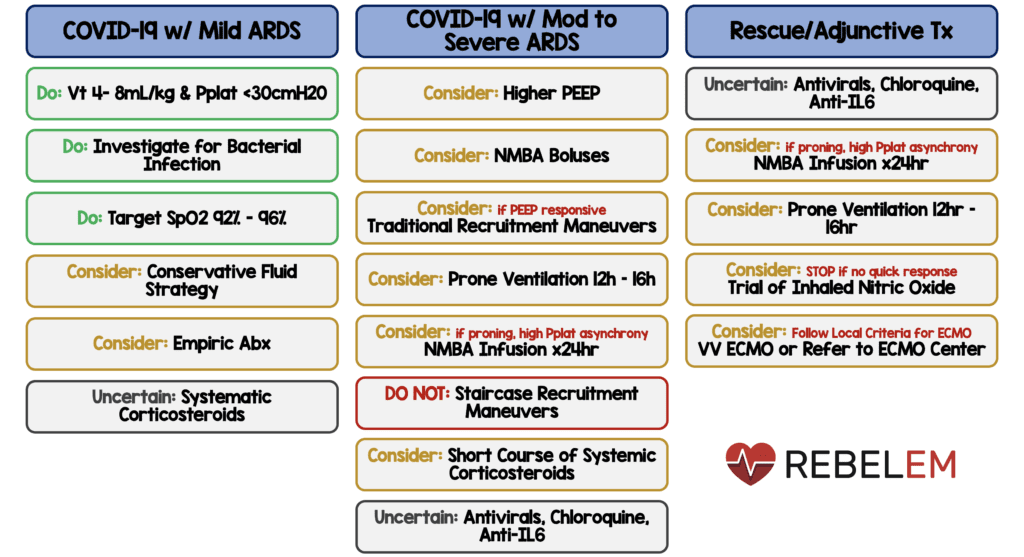
- ARDS = Low tidal volume (Vt) ventilation (Vt 4 – 8mL/kg of predicted body weight), > higher tidal volumes (Vt > 8mL/kg) (SR-MQE)
- ARDS = Target plateau pressure (Pplat) of <30cmH20 (SR-MQE)
- The ARDSNet study protocol set the initial Vt at 6mL/kg, and then measured Pplat (after a 0.5sec inspiratory pause). If the Pplat >30cmH20, Vt could be reduced in 1mL/kg (to 4mL/kg) steps until Pplat was within range
- Moderate to severe ARDS = Higher PEEP strategy, > lower PEEP strategy (WR-LQE)
- If using a higher PEEP strategy (i.e. PEEP >10 cmH20), clinicians should monitor patients for barotrauma
- ARDS = Conservative fluid strategy > liberal fluid strategy (WR-LQE)
- Moderate to severe ARDS = Prone ventilation for 12 to 16 hours > no prone ventilation (WR-LQE)
- Moderate to severe ARDS:
- Intermittent bolus neuromuscular blocking agents (NMBA) > continuous NMBA infusion (WR-LQE)
- Persistent ventilator dyssynchrony, need for ongoing deep sedation, prone ventilation, or persistently high plateau pressures = Continuous NMBA infusion for up to 48hrs (WR-LQE)
- ARDS = No routine use of inhaled nitric oxide (SR-LQE)
- Severe ARDS and hypoxemia despite optimizing ventilation and other rescue strategies = Trial of inhaled pulmonary vasodilator as a rescue therapy, if no rapid improvement in oxygenation is observed, the treatment should be tapered off (SR-VLQE)
- Hypoxemia despite optimizing ventilation = Recruitment maneuvers > not using recruitment maneuvers (WR-LQE)
- If recruitment maneuvers are used = Do not use staircase (incremental PEEP) recruitment maneuvers (SR-MQE)
- Refractory hypoxemia despite optimizing ventilation, with rescue therapies, and proning = Venovenous (VV) ECMO if available, or referring the patient to an ECMO center (WR-LQE)
COVID-19 Therapy:
- Mechanical ventilation + respiratory failure (without ARDS) = NO systemic corticosteroids (WR-LQE)
- Mechanical ventilation + ARDS = Consider systemic corticosteroids > not using corticosteroids (WR-LQE)
- Mechanical ventilation + respiratory failure = Empiric antimicrobials/antibacterial agents, over no antimicrobials (WR-LQE)
- If the treating team initiates empiric antimicrobials, they should assess for de-escalation daily, and re-evaluate the duration of therapy and spectrum of coverage based on microbiology results and the patient’s clinical status
- Fever = Acetaminophen/paracetamol for temperature control, over no treatment (WR-LQE)
- In critically ill adults with COVID-19:
- NO IVIG (Weak Recommendation, Very Low-Quality Evidence)
- NO convalescent plasma (WR-VLQE)
- No routine use of lopinavir/ritonavir (WR-LQE)
- Insufficient evidence for other antiviral agents in critically ill adults with COVID-19
- Insufficient evidence for recombinant rIFNs, alone or in combination with antivirals, in critically ill adults with COVID-19
- Insufficient evidence for chloroquine or hydroxychloroquine
- Insufficient evidence for tocilizumab
New Guideline Published
Surviving Sepsis Campaign: Guidelines on the Management of Critically Ill Adults with COVID19
Tweetorial on key clinical recommendations
#CCM #ICM #COVID19 @ESICM @SCCM @CritCareMed
— Ashley & Brooke Barlow PharmD (@theABofPharmaC) March 20, 2020
References:
- Alhazzani W et al. Surviving Sepsis Campaign: Guidelines on the Management of Critically Ill Adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med 2020. [Epub Ahead of Print]
- Loeb M et al. Surgical Mask vs N95 Respirator for Preventing Influenza Among Health Care Workers: Randomized Trial. JAMA 2009. PMID: 19797474
- CCH Leung et al. Comparison of High-Flow Nasal Cannula Versus Oxygen Face Mask for Environmental Bacterial Contamination in Critically Ill Pneumonia Patients: A Randomized Controlled Crossover Trial. J Hosp Infect 2019. PMID: 30336170
For More on This Topic Checkout:
- FOAMcast: Society of Critical Medicine Surviving Sepsis Guidelines #COVID19
- emDocs: Managing the Critical COVID-19 Patient – SCCM Guidelines
- First10EM: Surviving Sepsis Campaign COVID Guidelines
Post Peer Reviewed By: Anand Swaminathan, MD (Twitter: @EMSwami)
The post Surviving Sepsis Campaign Guidelines on the Management of Critically Ill Adults with COVID-19 appeared first on REBEL EM - Emergency Medicine Blog.


