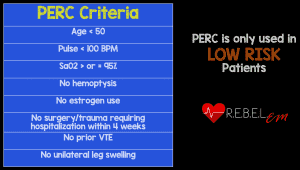
Background: The diagnosis of PE is a tricky thing. We want to limit over-testing patients and therefore, over-diagnosis. On the other hand, we don’t want to limit testing so much that we miss the diagnosis where treatment would make a difference. The pulmonary embolism rule-out criteria (PERC) was created to reduce testing in patients who have a very low probability of PE (i.e. prevalence of <1.8%) in which further testing would not be necessary. There have been many observational trials published on this score but until now there has not been a prospective randomized clinical trial (The PROPER Trial).
Background: The diagnosis of PE is a tricky thing. We want to limit over-testing patients and therefore, over-diagnosis. On the other hand, we don’t want to limit testing so much that we miss the diagnosis where treatment would make a difference. The pulmonary embolism rule-out criteria (PERC) was created to reduce testing in patients who have a very low probability of PE (i.e. prevalence of <1.8%) in which further testing would not be necessary. There have been many observational trials published on this score but until now there has not been a prospective randomized clinical trial (The PROPER Trial).
What They Did:
- Crossover cluster-randomized clinical non-inferiority trial in 14 EDs in France
- These were “low risk” clinical probability of PE patients being assessed for the safety of a PERC-based strategy
- PROPER Trial (PERC Rule to Exclude Pulmonary Embolism in the Emergency Department)
- Two, 6 month periods with a 2 month washout period before crossing over (i.e. half the EDs used PERC & half did not…after 2 months, the EDs that used PERC did not and vice versa):
- Control period (usual care)
- Age adjusted D-dimer testing and if positive à CTPA
- PERC based strategy (intervention period)
- If PERC = 0, then no more testing required
- If PERC ≥ 1 then usual diagnostic strategy applied
- CTPA with emboli were considered positive (i.e. including subsegmental PEs)
- If CTPA inconclusive further testing with VQ scan or Lower let Doppler US was performed
- Control period (usual care)
Outcomes:
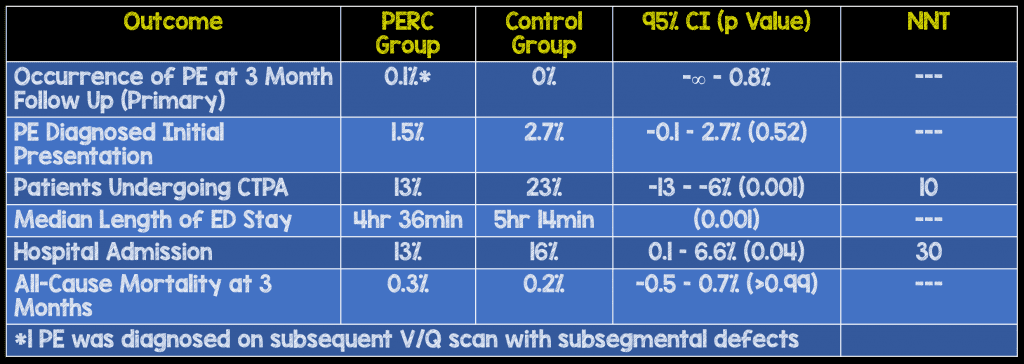
- Primary: Occurrence of thromboembolic event during 3 month follow up period, not diagnosed at original inclusion visit
- Secondary:
- Rate of CTPA
- Rate of CTPA adverse events requiring therapeutic intervention within 24hrs
- Median LOS in ED
- Rate of Hospital Admission or Readmission
- Onset of Anticoagulation
- Severe hemorrhage in patients with Anticoagulation
- All Cause Mortality at 3 months
Inclusion:
- All patients with suspicion of PE
- New onset presence or worsening SOB or chest pain and a low clinical probability of PE by physician gestalt as <15%
Exclusion:
- Obvious etiology other than PE (i.e. PTX or ACS)
- Acute severe presentation (Hypotension, SpO2<90%, Respiratory Distress)
- Contraindication to CTPA (Impaired renal function with CrCl <30mL/min, Known allergy to IV contrast)
- Pregnancy
- Inability to be followed up
- Already on anticoagulant therapy
Results:
- 1,916 patients included in analysis
- Mean age: 44 years
- PERC Group: 954 pts – 46 PERC negative patients (5%) underwent d-dimer testing and excluded from the analysis. It is unclear why they had additional testing
- Control Group: 954 pts
- 1,749 patients completed trial
Strengths:
- Multicenter, randomized clinical trial
- All patients instructed to return to the same ED or hospital with recurrent or worsening symptoms to help ensure no loss to follow up
- If unable to get in contact with patient for phone interview, patient’s general practitioner was contacted
- There was an adjudication committee of 3 hemostasis experts, who were blinded to the strategy allocation, that confirmed the occurrence of all suspected PEs and death due to PE
Limitations:
- The prevalence of PE was extremely low (i.e. 2.3%) in this study reducing the ability of this study to detect significant differences between groups
- This is a very young patient population (i.e. age 44 years) which may explain the low prevalence of PE in this study
- CTPA was defined as positive if showed isolated subsegmental PE, which is an issue because many of these could be left untreated
- With a less than 3% maximal failure rate in the control group, the calculated sample size was not accurate and would need to be larger
- Data on eligible patients who were not enrolled in this study were not available (i.e. 46 PERC negative patients who received d-dimer testing) making this a per-protocol analysis and not an intention to treat analysis
- 54 patients were lost to follow up and just a few PEs in these patients would have altered the conclusions of this study
Discussion:
- There were significantly more patients in the control group with a low Wells score of <2: 78% vs 91%
- Obviously, more d-dimer testing and CTPA in the control group: 50% vs 35% and 23% vs 13% respectively
Author Conclusion: “Among very low-risk patients with suspected PE, randomization to a PERC strategy vs conventional strategy did not result in an inferior rate of thromboembolic events over 3 months. These findings support the safety of PERC for very low-risk patients presenting to the emergency department.”
Clinical Take Home Point: In a “low risk” patient population, use of PERC over usual care, was non-inferior in both diagnosis and mortality associated with PE. An added benefit of using PERC over usual care in this study was a 10% decrease in imaging and 40min decrease in ED LOS.

References:
- Freund Y et al. Effect of the Pulmonary Embolism Rule-Out Criteria on Subsequent Thromboembolic Events Among Low-Risk Emergency Department Patients: The PROPER Randomized Clinical Trial. JAMA 2018. [Link HERE]
- Kline JA. Utiility of a Clinical Prediction Rule to Exclude Pulmonary Embolism Among Low-Risk Emergency Department Patients: Reason to PERC Up. JAMA 2018. [Link HERE]
For More Thoughts on This Topic Checkout:
- Ryan Radecki at EM Literature of Note: Using PERC & Sending Home Pulmonary Emboli For Fun and Profit
- Rory Spiegel at EMNerd (EMCrit): The Case of the Diagnostic Absurdity
- Scott Aberegg at PulmCCM: Ruling out PE in the ED – Critical Analysis of the PROPER Trial
- Salynn Boyles at PulmCCM: PERC Can Safely Rule Out Pulmonary Embolism in ED Setting
Post Peer Reviewed By: Anand Swaminathan (Twitter: @EMSwami)
The post Is it PROPER to PERC it Up? appeared first on REBEL EM - Emergency Medicine Blog.