
 Background: Publication of the RECOVERY trial results on Dexamethasone were game changing – the drug had a clear reduction in mortality in patients requiring oxygen. Since then, we have had little to celebrate in terms of therapeutics in those with moderate to severe disease. The beneficial effects of corticosteroids in COVID-19 patients with hypoxic lung damage suggests other, more specific immunomodulatory agents may provide additional patient-oriented improvements.
Background: Publication of the RECOVERY trial results on Dexamethasone were game changing – the drug had a clear reduction in mortality in patients requiring oxygen. Since then, we have had little to celebrate in terms of therapeutics in those with moderate to severe disease. The beneficial effects of corticosteroids in COVID-19 patients with hypoxic lung damage suggests other, more specific immunomodulatory agents may provide additional patient-oriented improvements.
Enter Tocilizumab. This is a recombinant anti-IL6 receptor monoclonal antibody that inhibits binding of IL-6 to receptors that signal inflammation. The results of tocilizumab from randomized trials thus far have shown mixed results for benefit. Many of the trials not showing benefit were smaller, however the larger REMAP-CAP trial [2] did report benefit in patients requiring organ support. Further data is clearly needed to guide clinicians.
Paper: RECOVERY Collaborative Group. Tocilizumab in Patients Admitted to Hospital with COVID-19 (RECOVERY): Preliminary Results of a Randomised, Controlled, Open-Label, Platform Trial. medRxiv Preprint 2021 [Link is HERE]
Clinical Question: Does the addition of tocilizumab to usual care in adult hospitalized patients with hypoxia and evidence of systemic inflammation improve 28d mortality compared to usual care alone?
What They Did:
- Randomized, controlled, open-label, platform trial
- RECOVERY: Randomised Evaluation of COVID-19 Therapy is assessing several treatments in patients hospitalized with COVID-19 in the UK
- This is an adaptive trial, that tests different treatment options vs usual care alone to see if it makes a difference in patient oriented outcomes. Once that is completed, the study moves on to the next treatment. In this case, tocilizumab became an option…
- Randomized in 1:1 ratio to:
- Usual standard care alone
- Tocilizumab
- IV over 60min + usual standard care
- >90kg = 800mg
- >65kg & ≤90kg = 600mg
- >40kg & ≤65kg = 400mg
- ≤40kg = 8mg/kg
- 2nd dose could be given 12 to 24hrs later if patient’s condition had not improved
Outcomes:
- Primary: 28d mortality
-
Secondary:
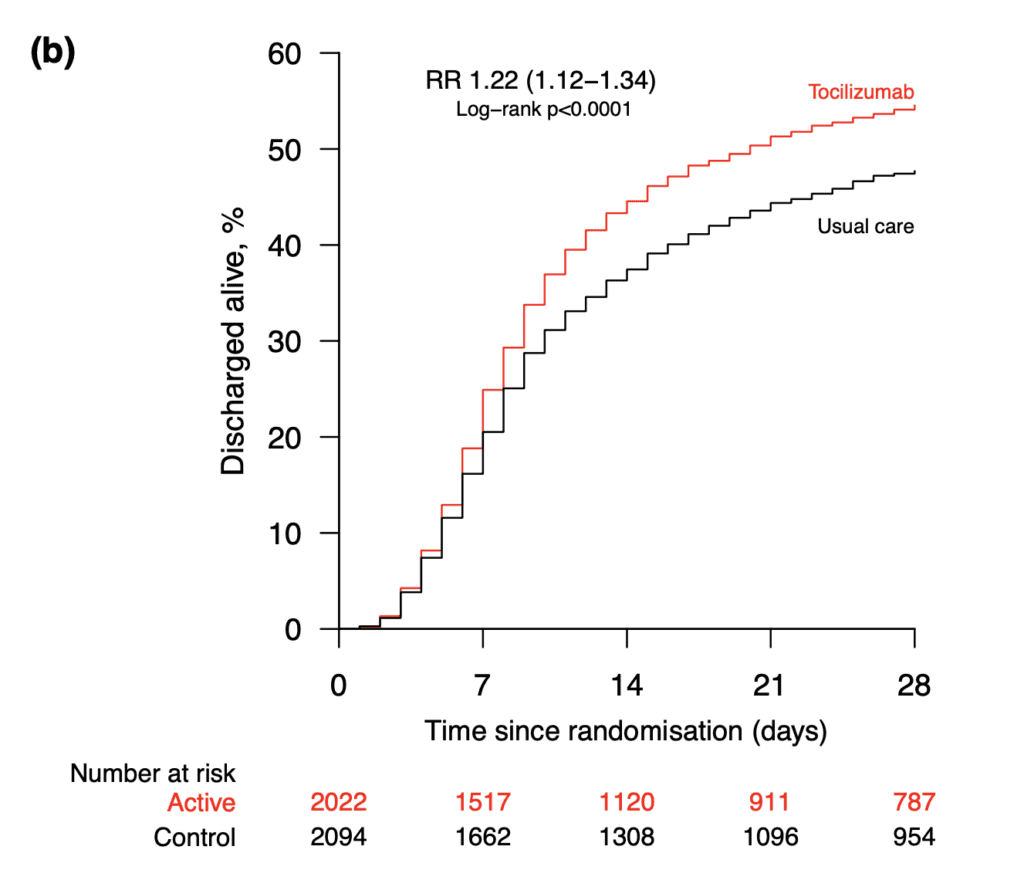
- Time to discharge alive from hospital
- Among patient not receiving invasive mechanical ventilation, receipt of invasive mechanical ventilation (including ECMO) and death
- Time to successful cessation of invasive mechanical ventilation (defined as cessation of invasive mechanical ventilation within, and survival to, 28d)
- Use of renal dialysis or hemofiltration
-
Safety:
- Cause-specific mortality
- Major cardiac dysrhythmia
Inclusion:
- Adult patients ≥18 years of age
- Clinically suspected or confirmed SARS-CoV-2 infection
- Hypoxia: O2 sat <92% on room air or requiring O2 therapy
- Evidence of systemic inflammation: CRP ≥75mg/L
Exclusion:
- Hypersensitivity to tocilizumab
- Evidence of active tuberculosis infection
- Clear evidence of active bacterial, fungal, viral or other infection (Besides COVID-19)
Results:
- 4116 adults included in the assessment of tocilizumab
- 562 (14%) were receiving invasive mechanical ventilation
- 1686 (41%) were receiving non-invasive respiratory support (HFNC, BPAP/CPAP)
- 1868 (45%) were receiving no respiratory support other than oxygen
- Number of days since symptom onset ≈9d
- 3385 (82%) of patients were receiving systemic corticosteroids at randomization
![]()
-
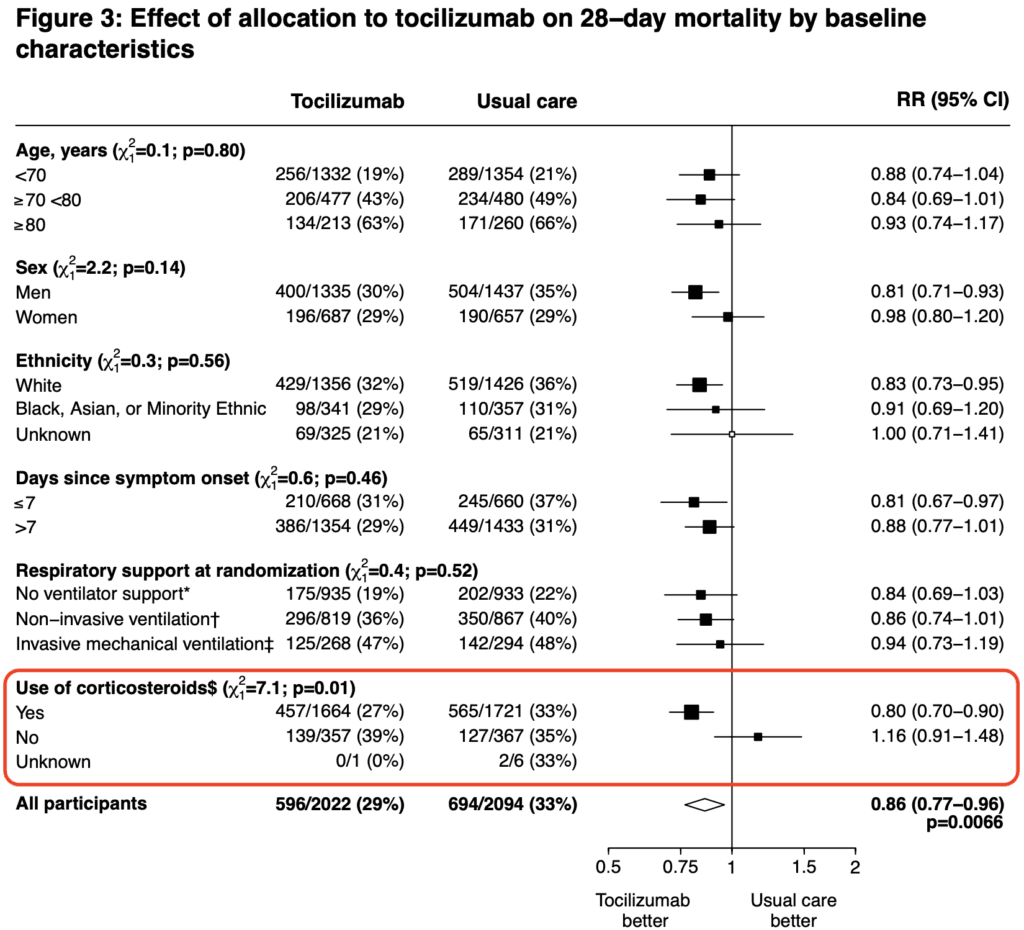
- Results were consistent in all pre-specified subgroups
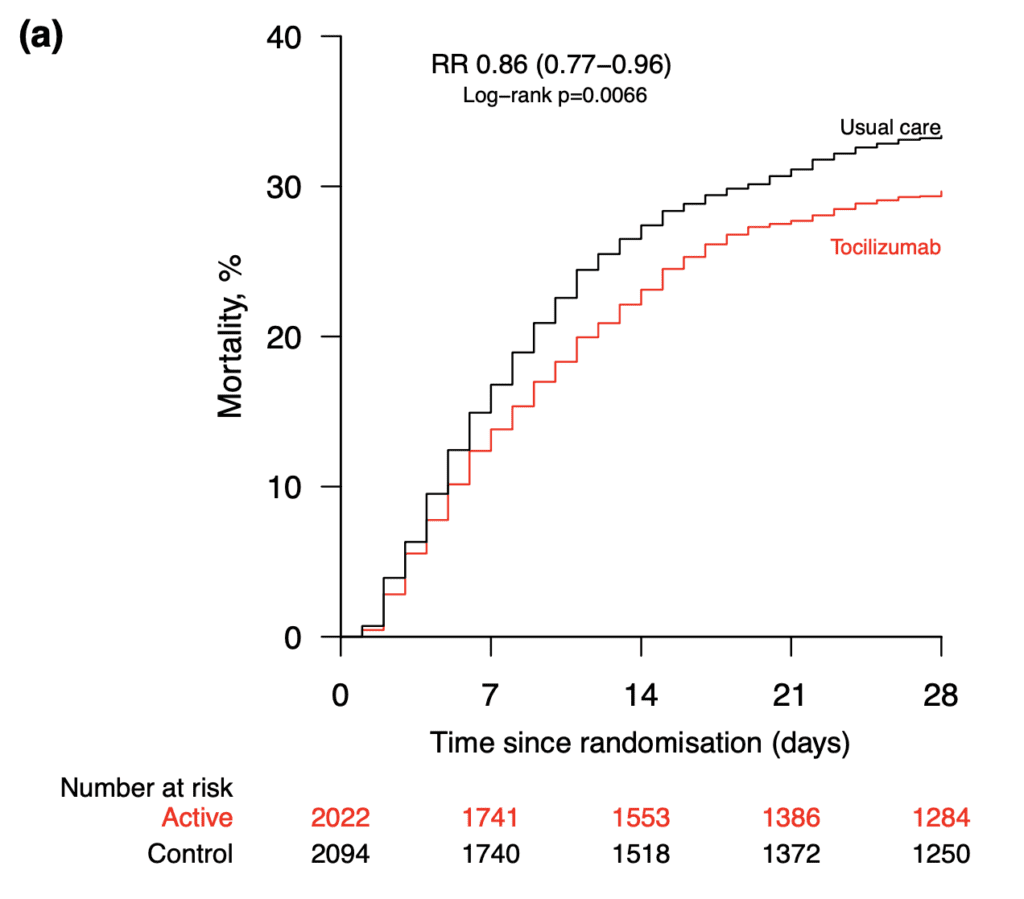
- An additional clear mortality benefit was seen in those receiving systemic corticosteroids (27% vs 33%; Risk Ratio 0.80; 95% CI 0.70 to 0.90)
- This mortality benefit was not seen in tocilizumab monotherapy (i.e. those not getting steroids)
![]()
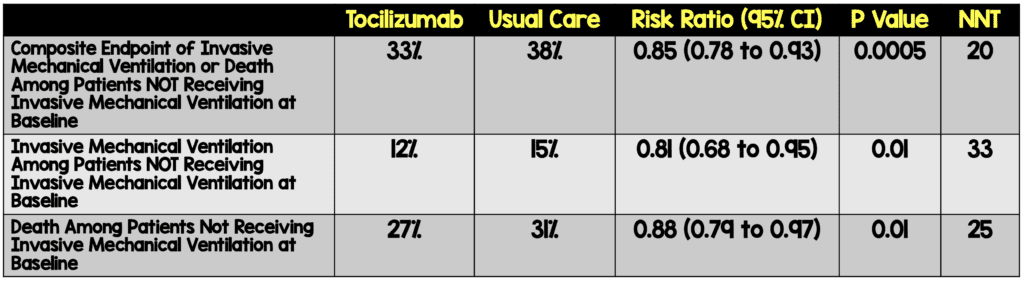
- Hemodialysis or Hemofiltration:
- Usual Care Alone: 7%
- Tocilizumab: 5%
- Risk ratio 0.75; 95% CI 0.59 to 0.96; p = 0.02
- No increase in cause-specific mortality from other infections as well as no difference in frequency of new cardiac arrhythmias
Strengths:
- Largest RCT on the use of tocilizumab + usual care vs usual care alone
- Tested combination of corticosteroids (standard care at this time) with tocilizumab
- Included patients requiring various levels of respiratory support (Simple O2 up to invasive mechanical ventilation)
- Neither the funders of the study nor Roche Products Ltd had any role in study design, data collection, data analysis, data interpretation, or writing of the manuscript
- Other treatments for COVID-19 were similar between groups
- Benefits of primary outcome was consistent across all pre-specified patient groups increasing validity of results found
Limitations:
- For this preliminary report the primary outcome results are available for 92% of randomized patients
- Open-label design which could affect subjective outcomes, however most likely no major effect on objective outcomes such as mortality
- Tocilizumab will not be available at all hospitals as it is expensive and production may not be able to keep up with the demand
- 17% of patients in the tocilizumab group did not receive the drug and the reasons for this were not recorded
- Hospital stay for many patients is >28days and therefore we do not have long-term outcomes, but there is a pre-planned analysis at 6 months that will provide this information
- It is unclear in patients with CRP <75mg/L if there would be a benefit from tocilizumab
Discussion:
- The authors state there are 7 previous RCTs on the use of tocilizumab for the treatment of COVID-19. Six of these had fewer than 100 deaths in each trial whereas the REMAP-CAP trial recruited critically ill patients of which >99% of them required non-invasive respiratory support or invasive mechanical ventilation. Taken together the trials did not show a significant mortality benefit (death rate ratio 0.91; 95% CI 0.72 to 1.14). The RECOVERY trial has 3x as many deaths as all the previous trials combined. The addition of the RECOVERY trial shows a that tocilizumab is associated with a 13% reduction in 28d mortality (death rate ratio 0.87; 95% CI 0.79 to 0.96; p = 0.005)
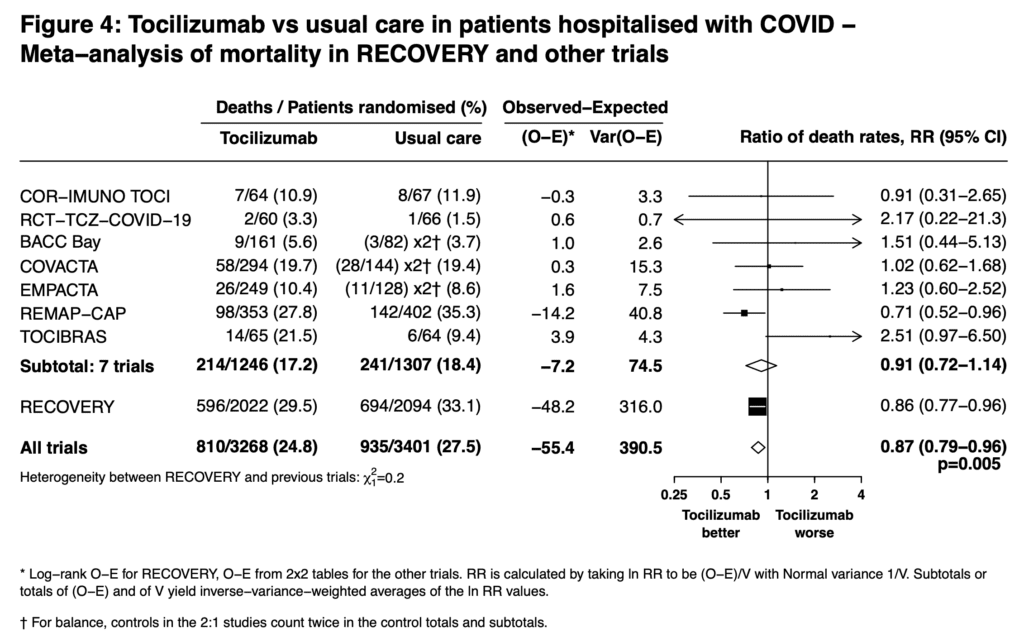
- Sarilumab is another IL-6 antagonist which was not studied in this trial and in the REMAP-CAP trial only 48 patients received it, therefore we cannot make any conclusions on this medication
- Tocilizumab costs about $2400 per infusion [drugs.com]
-
Potential Workflow for Admitted COVID-19 patients:
- COVID-19 patient requiring hospitalization + O2 Requirement = Corticosteroids
- COVID-19 patient requiring hospitalization + O2 Requirement + CRP ≥75mg/L = Corticosteroids + Tocilizumab
Author Conclusion: “In hospitalized COVID-19 patients with hypoxia and systemic inflammation, tocilizumab improved survival and other clinical outcomes. These benefits were seen regardless of the level of respiratory support and were additional to the benefits of systemic corticosteroids.”
Clinical Take Home Point: In hospitalized patients with COVID-19 pneumonia who are hypoxic (O2 sat <92% on RA or requiring O2 therapy) and have evidence of systemic inflammation (CRP ≥75mg/L), tocilizumab, when combined with corticosteroids, reduces mortality. Additionally, there is an association with increased chance of successful hospital discharge and reduced chance of requiring invasive mechanical ventilation compared to usual care alone. Tocilizumab + Corticosteroids should now be standard care, where available, in patients who meet criteria.
References:
- RECOVERY Collaborative Group. Tocilizumab in Patients Admitted to Hospital with COVID-19 (RECOVERY): Preliminary Results of a Randomised, Controlled, Open-Label, Platform Trial. medRxiv Preprint 2021 [Link is HERE]
- REMAP-CAP Investigators. Interleukin-6 Receptor Antagonists in Critically Ill Paitents with COVID-19 – Preliminary Report. medRxiv 2021. [Link is HERE]
For More Thoughts on This Topic Checkout:
- The Bottom Line: REMAP-CAP IL-6
- PulmCrit: Six RCTs to Answer One Question – What is the Role of Tocilizumab in COVID-19?
- PulmCrit: RECOVERY Confirms Benefit of Toci Combined with Dexamethasone
- FOAMCast: Tocilizumab in COVID-19 – The RECOVERY Trial
Post Peer Reviewed By: Anand Swaminathan, MD (Twitter: @EMSwami)
The post The RECOVERY Trial: Tocilizumab appeared first on REBEL EM - Emergency Medicine Blog.


