 Corneal abrasions account for 10% of all ocular complaints, and are the most common cause of ocular trauma (Alotaibi 2011, Bhatia 2013). The diagnosis of corneal abrasions typically involves fluorescein staining of the eye and visualization of the abrasion via slit lamp exam. This review focuses specifically on pain control for corneal abrasions.
Corneal abrasions account for 10% of all ocular complaints, and are the most common cause of ocular trauma (Alotaibi 2011, Bhatia 2013). The diagnosis of corneal abrasions typically involves fluorescein staining of the eye and visualization of the abrasion via slit lamp exam. This review focuses specifically on pain control for corneal abrasions.
Although corneal abrasions typically heal within 24-72 hours without complications, the pain in the acute phase is usually significant (Wilson 2004). Treatments described include patching, topical anesthetics, topical NSAIDS, cycloplegics or oral analgesics. Utilization of topical anesthetics has been described in a previous post. Oral analgesics are usually prescribed as a rescue modality when topical treatment is ineffective at managing pain.
Patching
Patching used to be standard of care. The reasoning for its use was that it would reduce blinking and corneal trauma caused by eyelid closure thereby reducing pain (Thiel 2017). However, it was found that patients have poorer outcomes with patching. This is due to decreased oxygen delivery to the eye and higher risks of infection due to increased moisture (Thiel 2017).
Cycloplegics
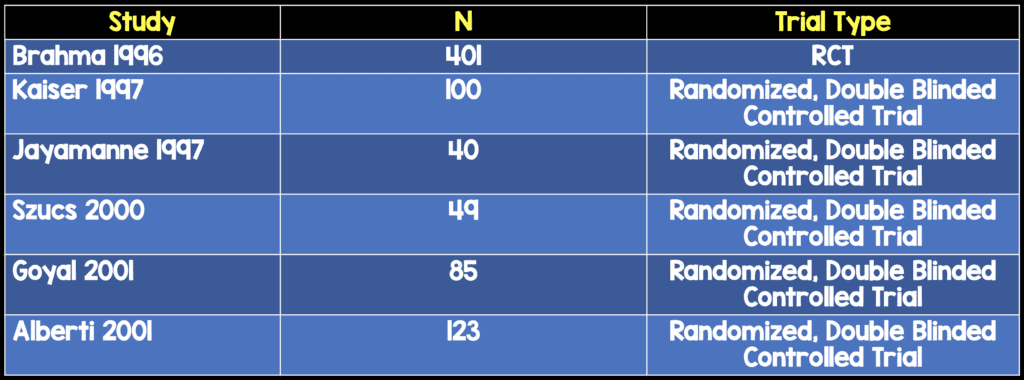
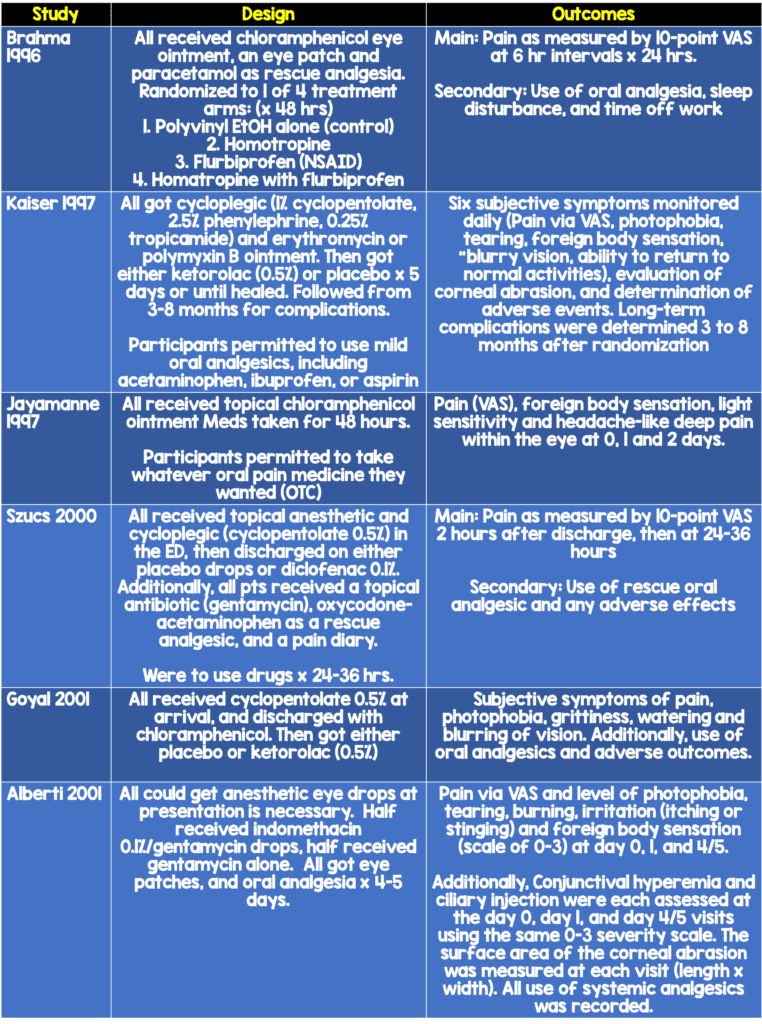
Some textbooks report that cycloplegics may be considered as an adjunct for pain control due to causing relief from ciliary muscle spasm, photophobia, and blepharospasm (Bhatia 2013, Butler 2017). However, review of the literature demonstrates that the evidence behind this practice is sparse and does not support its widespread use. A study of 40 patients who were randomized to receive 5% homotropine vs placebo found no differences in pain scores (Meek 2010). Another study of 401 patients randomized to lubrication alone vs 2% homatrapine (single dose) vs flubiprofen 0.03% QDS vs flubiprofen 0.03% qds and homatropine found no difference between homatropine and other treatments (Brahma 1996).
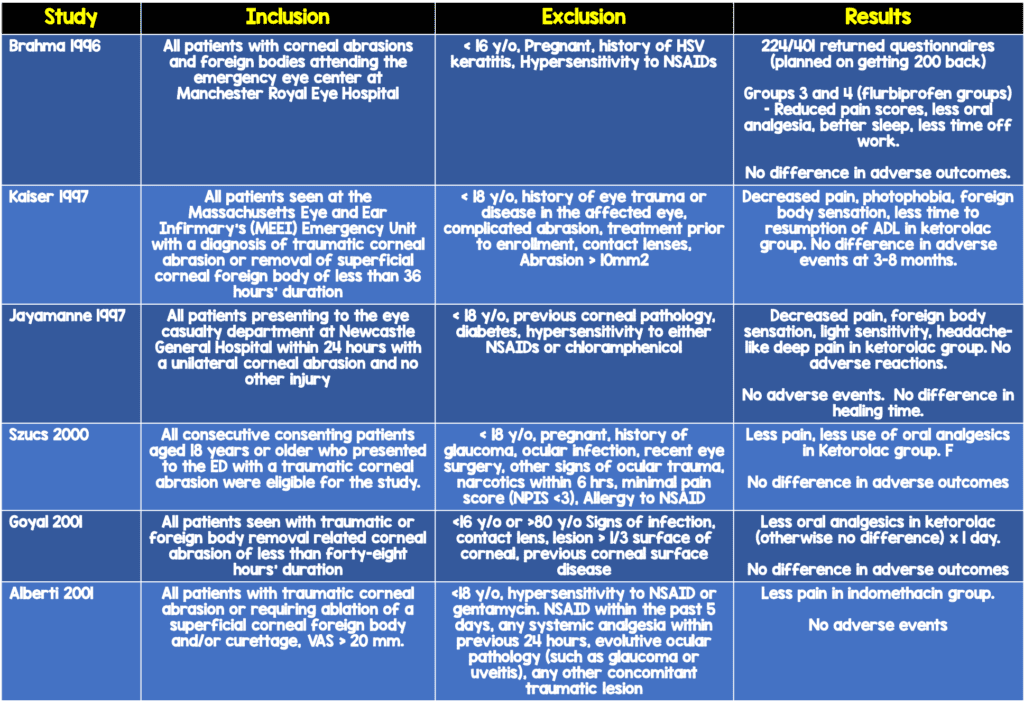
Topical NSAIDS
In the year 2000, there were around 200 cases of corneal melting described by 106 physicians associated with a specific generic form of topical ketorolac (Flach 2000). Although that product was removed from the market, there still remains reluctance to use topical NSAIDS due to the potential for serious toxicity. Since then, there have been a few case reports and case series that do mention an association with topical NSAIDS and corneal pathology. However, these cases are in mostly in post ocular surgery patients, and include patients who are on the topical NSAIDS for much longer than is recommended for corneal abrasion treatment

There have been multiple randomized controlled trials that have demonstrated, however, that topical NSAIDS used for corneal abrasions for 1-3 days are not associated with any adverse outcomes
Clinical Bottom Line:
In summary, patching can lead to harm and should be avoided, while cycloplegics have not been shown to help with pain. Although topical treatment with anesthetics and NSAIDS have traditionally been avoided due to concern for worse outcomes, multiple placebo-controlled randomized controlled trials have demonstrated better patient outcomes without any increased incidence of adverse events in uncomplicated traumatic abrasions presenting to the emergency department if used for only 24-36 hours.
Guest Contributor

References:
- Alotaibi AG, Osman EA, Allam KH, Abdel-Rahim AM, Abu-Amero KK. One month outcome of ocular related emergencies in a tertiary hospital in Central Saudi Arabia. Saudi medical journal. 2011; 32(12):1256-60. [pubmed]
- Bhatia, K., & Sharma, R. (2013). Chapter 26: Eye Emergencies. In Emergency Medicine Clinical Essentials(2nd ed., pp. 209-225). Philadelphia: Elsevier.
- Wilson SA, Last A. Management of corneal abrasions. American family physician. 2004; 70(1):123-8. [pubmed]
- Flach A. Topically applied nonsteroidal anti-inflammatory drugs and corneal problems: an interim review and comment. Ophthalmology. 2000; 107(7):1224-6. [pubmed]
- Lin JC, Rapuano CJ, Laibson PR, Eagle RC, Cohen EJ. Corneal melting associated with use of topical nonsteroidal anti-inflammatory drugs after ocular surgery. Archives of ophthalmology (Chicago, Ill. : 1960). 2000; 118(8):1129-32. [pubmed]
- Guidera AC, Luchs JI, Udell IJ. Keratitis, ulceration, and perforation associated with topical nonsteroidal anti-inflammatory drugs. Ophthalmology. 2001; 108(5):936-44. [pubmed]
- Isawi H, Dhaliwal DK. Corneal melting and perforation in Stevens Johnson syndrome following topical bromfenac use. Journal of cataract and refractive surgery. 2007; 33(9):1644-6. [pubmed]
- Bekendam PD, Narváez J, Agarwal M. Case of corneal melting associated with the use of topical nepafenac. Cornea. 2007; 26(8):1002-3. [pubmed]
- Di Pacuale MA. Corneal Melting After Use of Nepafenac in a Patient with Chronic Cystoid Macular Edema. Eye Contact Lens 2008; 34(2): 129 – 30. [pubmed]
The post Topical Pain Control for Corneal Abrasions appeared first on REBEL EM - Emergency Medicine Blog.
