
 Background: Awake prone positioning was an intervention adopted early in the COVID-19 pandemic for patients with hypoxemic respiratory failure. Unfortunately, many early trials were not powered to show improvements in oxygenation, escalation of respiratory support, or mortality. The one meta-analysis (Link is HERE) including 50 trials with ≈3000 patients suffered from methodological flaws of the included studies. Most of the trials were small, observational, with very wide confidence intervals which made interpreting these trials of limited utility and really an interpretation of the absence of evidence, rather than an absence of benefit. Despite the lack of high-quality evidence, many clinicians continue to apply awake prone positioning and the approach has even incorporated into some clinical guidelines. Better evidence to guide this practice is clearly needed.
Background: Awake prone positioning was an intervention adopted early in the COVID-19 pandemic for patients with hypoxemic respiratory failure. Unfortunately, many early trials were not powered to show improvements in oxygenation, escalation of respiratory support, or mortality. The one meta-analysis (Link is HERE) including 50 trials with ≈3000 patients suffered from methodological flaws of the included studies. Most of the trials were small, observational, with very wide confidence intervals which made interpreting these trials of limited utility and really an interpretation of the absence of evidence, rather than an absence of benefit. Despite the lack of high-quality evidence, many clinicians continue to apply awake prone positioning and the approach has even incorporated into some clinical guidelines. Better evidence to guide this practice is clearly needed.
Paper: Ehrmann S et al. Awake Prone Positioning for COVID-19 Acute Hypoxaemic Respiratory Failure: A Randomised, Controlled, Multinational, Open-Label Meta-Trial. Lancet Respir Med 2021. PMID: 34425070[Access on Read by QxMD]
Clinical Question: Does an awake prone position help prevent intubation or death in patients with severe COVID-19?
What They Did:
- Prospective, collaborative meta-trial of six randomized controlled open-label superiority trials of adults who required respiratory support with high-flow nasal cannula for acute hypoxemic respiratory failure due to COVID-19
- Meta-trial = Incorporating a collaborative prospective meta-analysis of individual patient data from each randomized controlled trial
- Hospitals from six countries were involved including Canada, France, Ireland, Mexico, USA, and Spain
- Patients were randomized to:
- Awake prone positioning
- Instructed and assisted to lie in prone position for as long and as frequently as possible each day
- HFNC initiated at maximally tolerated flow setting and FiO2 titrated to maintain SpO2 between 90 and 95%
- Use of non-invasive ventilation was not included in trial protocol but was recorded prospectively
- Standard care
- HFNC initiated at maximally tolerated flow setting and FiO2 titrated to maintain SpO2 between 90 and 95%
- Use of awake prone position as a rescue intervention was discouraged and recorded as a protocol violation
- Awake prone positioning
Outcomes:
-
Primary: Treatment failure within 28 days
- Treatment failure = proportion of patient intubated or dying within 28 days
- Trigger to intubate:
- Worsening respiratory failure (RR >40BPM)
- Respiratory muscle fatigue
- Respiratory acidosis (pH <7.25)
- Copious tracheal secretions
- Severe hypoxemia with SpO2 <90% despite an FiO2 of ≥0.8
- Hemodynamic instability
- Deteriorating mental status
-
Secondary:
- Intubation
- Mortality
- Use of non-invasive ventilation
- Length of hospital stay
- Time to high flow nasal cannula weaning in patients with treatment success
- Defined as patient being alive and not having required intubation within 28 days of enrollment
- Time to treatment failure
- Time to intubation
- Time to death
- Duration of invasive mechanical ventilation in intubated patients surviving to day 28
- Mortality in invasively mechanically ventilated patients
-
Safety:
- Ratio of SpO2/FiO2 to respiratory rate (i.e ROX index)
Inclusion:
- >18 years of age
- Acute hypoxemic respiratory failure due to proven (or highly clinically suspected, pending microbiological confirmation) COVID-19 pneumonia
- Acute hypoxemic respiratory failure = Respiratory support with HFNC and a ratio of SpO2 to FiO2 of ≤315mmHg (This is equivalent to PaO2 to FiO2 ratio of ≤300mmHg)
Exclusion:
- Patients unable or refused to provide informed consent
- Hemodynamically unstable
- Severely obese with BMI >40kg/m2
- Pregnancy
- Contraindication to awake prone positioning
Results:
- 1126 patients enrolled and randomized
- 5 patients withdrew from the study leaving 1121 patients for analysis
- Average age = 61 years
- Average BMI = 29.7kg/m2
- Confirmed COVID-19 = 99%
- Use of Steroids = 88%
- Hospital location at enrollment:
- ICU = 60%
- Intermediate Care Unit = 35%
- ED = 1%
- General Ward = 4%
- Top Countries of recruitment:
- Mexico: 38%
- France: 36%
- USA: 20%
- Median Daily Duration of Awake Prone Positioning = 5hrs (Range 1.6 to 8.8hrs)
-
Treatment Failure within 28d (Intubation or Death):
- Awake Prone Positioning: 223/564 (40%)
- Standard Care: 257/557 (46%)
- RR 0.86; 95% CI 0.87 to 0.98; p = 0.02
- NNT = 16.7
- Intubation Rate at Day 28:
- Awake Prone Positioning: 33%
- Standard Care: 40%
- NNT = 14
- Mortality at Day 28
- Awake Prone Positioning: 21%
- Standard Care: 24%
- RR 0.87; 95% CI 0.71 to 1.07
- Rest of the Secondary Outcomes
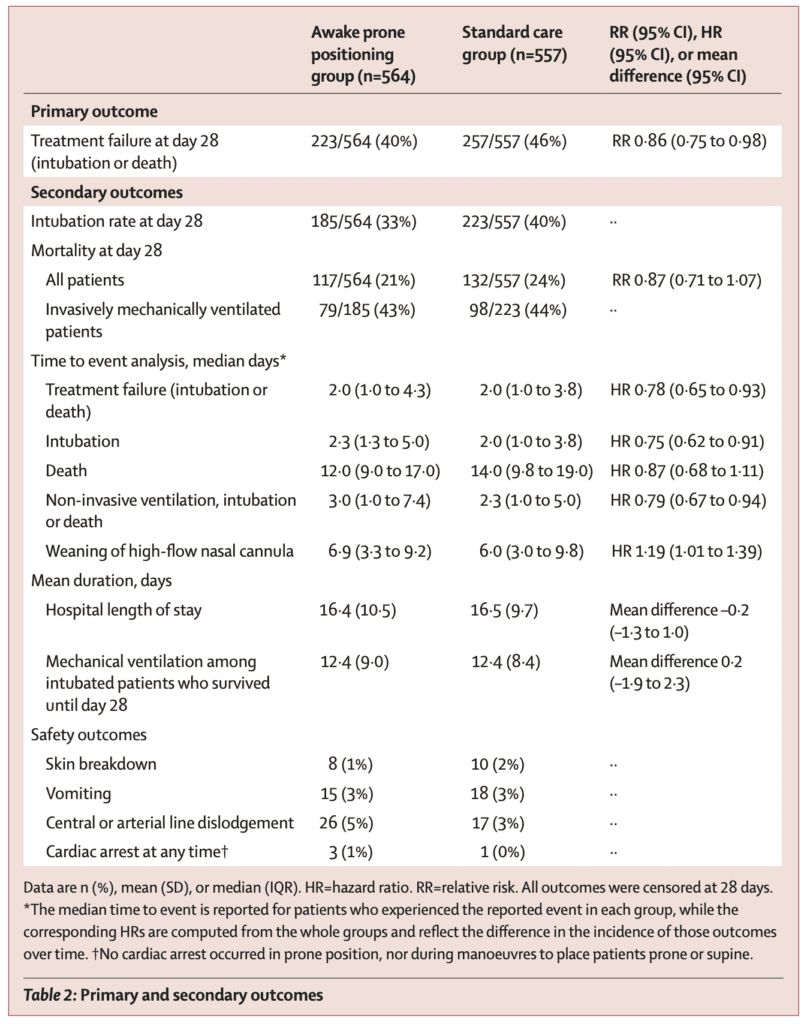
- Physiologic Effects of Awake Prone Positioning
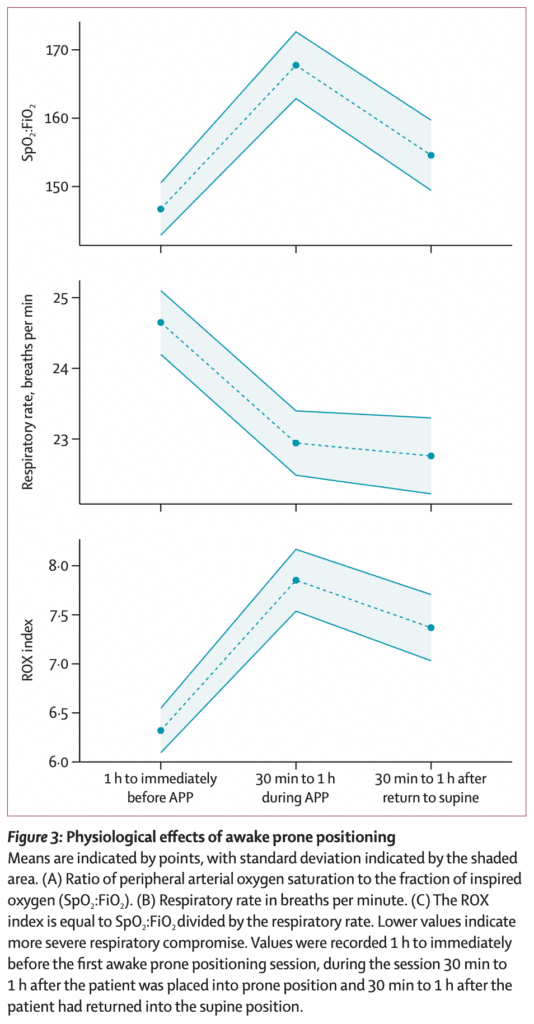
- Incidence of prespecified adverse events were low and similar in both groups (Skin breakdown, vomiting, central line or a-line dislodgement)
Strengths:
- Highest quality evidence to date on the use of awake prone positioning during the COVID-19 pandemic
- Large sample size and international inclusion increases generalization of results to a variety of settings
- Harmonized inclusion, exclusion, planned intervention, and a common set of core data to be extracted from each trial
- Primary and secondary outcomes were agreed upon prior to trial initiation with a collaborative reporting of results as a unified group
- Meta-trial approach combined a prospective design, and the high power of a large multinational trial with the convenience of a faster setup time and lower cost compared with a centralized international trial
- Cumulative sample size of several trials, provides adequate power for most effect sizes that are difficult to estimate during a pandemic
- Meta trial also allows for coordinated prospective interim analysis which can lead to reduced time to reach a conclusion compared with individually conducted trials
- As the decision to intubate is a subjective one, an objective set of criteria was created to harmonize triggers for intubation
- 99% of patients randomized completed the study and included in the analysis (Only 5 patients withdrew consent)
- Baseline characteristics of included patients were balanced between groups (Including baseline demographics, disease characteristics, and SpO2:FiO2)
Limitations:
- Physicians were unblinded to treatment which could cause bias. Intubation a subjective outcome can be affected by unblinding, whereas mortality an objective outcome would be less likely to be affected by blinding
- Subsequent management after intubation (including prone positioning) was left at the discretion of the treating physician
- In the standard care group one out of every ten patients underwent awake prone position. This would lead to an underestimation of the efficacy of awake prone position
- Patients with BMI > 40 were excluded (likely proning difficult in this population). Most likely need special mattresses or pillows to assist in this population
- Only included patients on HFNC. If needed CPAP/BPAP couldn’t enter into the trial. This may have removed a group that can really benefit
- Granular detail on how long patients were proning not given
Discussion:
- Mortality and duration of invasive mechanical ventilation were similar between groups among intubated patients. This suggest there is no signal for harm from awake prone positioning (i.e. P-SILI)
- The effect size of awake prone positioning on the primary outcome was greatest in the trial from Mexico, which also had the longest mean daily duration of awake prone positioning. Longer awake prone positioning throughout the day was associated with greater treatment success
- Treatment Failure:
- Awake Prone Position on HFNC at Least 8h Daily on Average (151pts) = 17%
- Awake Prone Position on HFNC <8h Daily on Average (413pts) = 48%
- As many patients cannot tolerate long periods of time for awake prone positioning, more effort should be placed into modifiable factors that will help promote this intervention
- Treatment Failure:
Author Conclusion: “Awake prone positioning of patients with hypoxaemic respiratory failure due to COVID-19 reduces the incidence of treatment failure and the need for intubation without any signal of harm. These results support routine awake prone positioning of patient with COVID-19 who require support with high-flow nasal cannula.”
Clinical Take Home Point: This is the highest-level evidence we have to date on awake prone positioning. In this trial of patients with hypoxemic respiratory failure due to COVID-19, awake prone positioning reduced the incidence of treatment failure (intubation and death), which was primarily driven by decreased intubation compared to standard care. Not only was there no signal of harm from awake prone positioning, but even longer durations of awake prone positioning (>8hrs/day) were associated with a lower risk of treatment failure.
References:
- Ehrmann S et al. Awake Prone Positioning for COVID-19 Acute Hypoxaemic Respiratory Failure: A Randomised, Controlled, Multinational, Open-Label Meta-Trial. Lancet Respir Med 2021. PMID: 34425070 [Access on Read by QxMD]
For More Thoughts on This Topic Checkout:
- REBEL EM: COVID-19 Awake Proning – All Hype?
Post Peer Reviewed By: Anand Swaminathan, MD (Twitter: @EMSwami)
The post Awake Proning for COVID-19 Acute Hypoxemic Respiratory Failure appeared first on REBEL EM - Emergency Medicine Blog.
