
 Background: One of the hot topics in COVID-19 care is the mortality rate associated with invasive mechanical ventilation (IMV). There have been early reports of IMV having mortality rates ranging from 50 to 90%. These high rates are concerning but, context is important; many of the reports emerged from areas with large surges where hospital systems were overwhelmed. Additional data looking at outcomes of critical patients is important particularly within systems that were able to maintain baseline critical care provisions despite surges.
Background: One of the hot topics in COVID-19 care is the mortality rate associated with invasive mechanical ventilation (IMV). There have been early reports of IMV having mortality rates ranging from 50 to 90%. These high rates are concerning but, context is important; many of the reports emerged from areas with large surges where hospital systems were overwhelmed. Additional data looking at outcomes of critical patients is important particularly within systems that were able to maintain baseline critical care provisions despite surges.
Paper: Auld SC et al. ICU and Ventilator Mortality Among Critically Ill Adults with COVID-19. Critical Care Medicine 2020. [Epub Ahead of Print]
Clinical Question: What is the associated risk of death in patients admitted to the ICU, with COVID-19 pneumonia, requiring invasive mechanical ventilation within a 3 hospital system in Georgia?
What They Did:
- Multicenter, retrospective, observational cohort study of patients admitted to the ICU
- 6 COVID-19 designated ICUs from three hospitals in Atlanta
- Included:
- Adults ≥18 years of age
- Confirmed severe COVID-19
- Admitted to ICU
Results:
- 217 patients included in analysis
- Median age was 64 years with 49 patients (22.6%) >75 years of age
- Majority of patients were African American (70.5%)
- HTN was the most common comorbid condition (61.7%)
- DM was the second most common comorbid condition (45.6%)

Discussion:
- Patients seemed to be in the inflammatory stage of disease with a median d-dimer 1731ng/mL (upper limit of normal 298ng/mL), and elevated CRP 190mg/L (upper limit of normal 10mg/L).
- Clinically, this was a sick cohort of patients with a median SOFA score of 7 (predicted mortality of 21.5%), PaO2/FiO2 ratio of 132 (range 100 – 178), a median static lung compliance on the 1st day of intubation of 34mL/cmH2O (range 28 to 46mL/cmH2O), 65.9% requiring vasopressor support for shock, and 29.0% requiring renal replacement therapy.
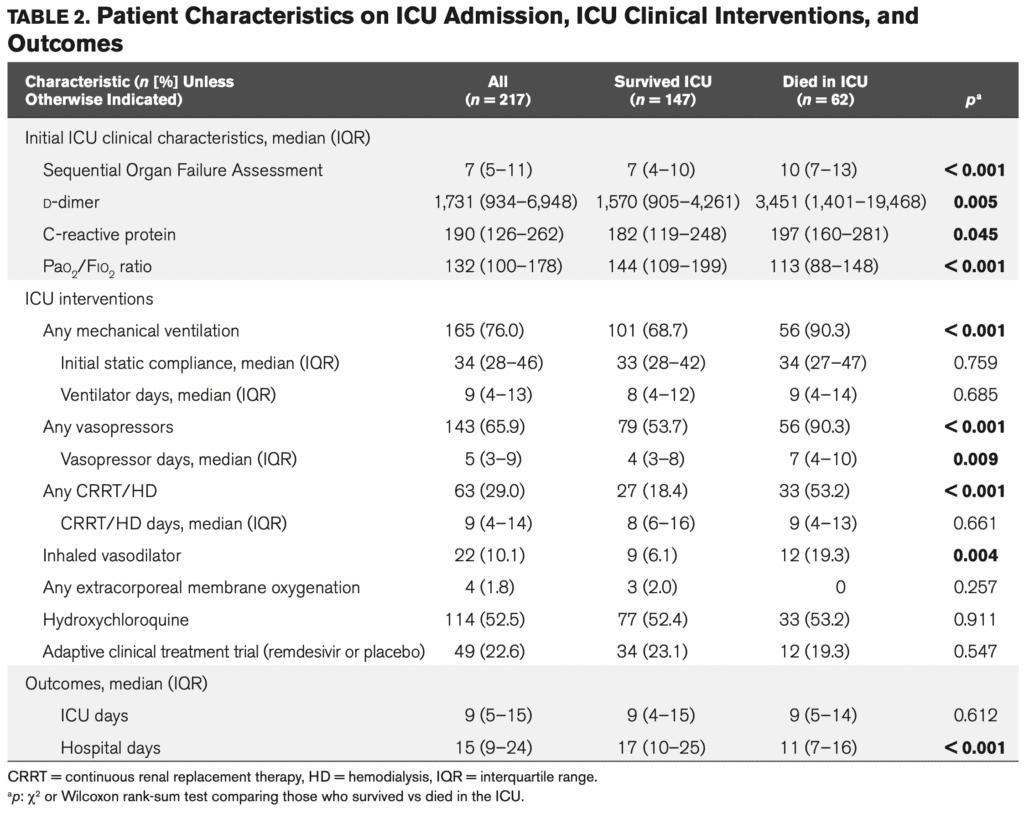
Patient Characteristics on ICU Admission, Interventions, and Outcomes
- Medications used during this trial:
- Inhaled pulmonary vasodilators: 10.1%
- At least one dose of hydroxychloroquine: 52.5%
- Remdesivir: 22.6%
- Authors state there was no difference in survival in patient taking hydroxychloroquine or remdesivir (I assume this is in comparison to placebo for remdesivir as the ACTT trial was an RCT, but unsure which trial they are referring to for hydroxychloroquine)
- The total duration of recruitment was from March 6th to April 17th, 2020 (42 days). 217 patients were recruited during this time (This averages out to ≈5 patients per day admitted to the ICU with COVID-19). This was ≈5/day spread over 6 ICUs in addition, which is not even 1 patient/day for all the ICUs. Patients were able to be rapidly brought to the ICU and had standard ICU ratios and teams (i.e. they were never overwhelmed). For comparison, the admission rates in New Jersey at Swami’s shop were 10/shift to the ICU in the first 7 to 10 days of the surge.
Author Conclusion: “Despite multiple reports of mortality rates exceeding 50% among critically ill adults with coronavirus disease 2019, particularly among those requiring mechanical ventilation, our early experience indicates that many patients survive their critical illness.”
Clinical Take Home Point: How do we reconcile a study where the “standard” protocol used emphasized “early” intubation and “standard” lung protective ventilation strategies to previous studies showing higher mortality rates? The devil is in the details…
- Study occurred later in the pandemic: Allowed preparation of the system with organizational structures, equipment, and consensus-driven protocols
- There was no shortage of ICU beds: Patient care was conducted in the ICU by a critical care team with experience in managing acute respiratory failure
- 217 Patients in 42 Days: Lower nurse and physician/patient ratios
- No shortage of medications or critical care equipment
References:
- Auld SC et al. ICU and Ventilator Mortality Among Critically Ill Adults with COVID-19. Critical Care Medicine 2020. [Epub Ahead of Print]
Post Peer Reviewed By: Anand Swaminathan, MD (Twitter: @EMSwami)
The post COVID-19: IMV and Mortality at Emory appeared first on REBEL EM - Emergency Medicine Blog.


